This is default featured slide 1 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 2 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 3 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 4 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 5 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
Saturday, 13 February 2016
Branches of Chemistry together with the insight
Dalton's Atomic Theory
The reaction equated with Method Ping pong
Mol concepts
TOPICS: Concepts Mol
Mol concepts
If in our daily lives we know the unit number in the form of a dozen (12 pieces), scores (20 pieces), and the rim (500 pieces) then the chemistry we know the mole as the unit number. One mole is defined as the amount of a substance containing particles according to the number of atoms in 12 grams of carbon-12. Since the mass of one atom C-12 is 1.992 × 10-23 grams, then the number of atoms in one mole (12 grams) of C-12 can be calculated as follows:
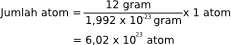
the number of atoms
Numbers 6.02 x 1023 is called Avogadro's number and Latau denoted by NA. Thus, one mole of a substance is the amount of substance containing 6.02 x 1023 particles. If n states the number of moles and N denotes the number of atoms or molecules, then the relationship n and N can be written in the following formula.
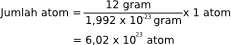
Molar mass
The molar mass is the mass of one mole of a substance. The value of the molar mass of a substance is always the same with Ar or Mr substances. The relationship of mass and number of moles of a substance can be written in the form of the following formula.
gram-mole
Volume Molar Gas
Gas molar volume is the volume of 1 mol of gas. Due to the volume of gas is strongly influenced by temperature and pressure, the stoichiometry of the chemists define a reference condition in the determination of the molar volume. This reference condition is 0 ° C (273 K) and 1 atm. This condition is called standard conditions or STP (Standard Temperature and Pressure). At STP conditions, molar volume of gas is 22.4 L. Thus, the relationship number of moles of gas (n) with its volume can be written by the following equation.
volume-mol
Volume (V) and the molar volume (Vm) gas expressed in liters.
- See more at: http://apapikir.com/2016/02/12/konsep-mol/#sthash.AxJOz9xS.dpuf
The relative atomic mass and molecular mass Relative
TOPICS: relative atomic mass and molecular mass Relative
The relative atomic mass and molecular mass Relative
Source: http: //www.mystupidtheory.com/
Atoms are very small particles. Because the atomic mass is also too small when expressed in units of grams so that the chemists created a way to measure the mass of an atom, namely the relative atomic mass. The relative atomic mass (Ar) is the ratio of the average mass of an atom with one-twelfth times the mass of one atom of carbon-12.
Ar
The smallest unit of a substance can also be a molecule. Molecules composed by two or more atoms held together by chemical bonds. Relative molecular mass (Mr) adalahperbandingan average mass of a molecule with one-twelfth times the mass of one atom of carbon-12.
Mr
In the above formulas used atomic mass and molecular mass on average. Why use an average atomic mass? Because the elements in nature have beberapaisotop. For example, the carbon in nature has two pieces of stable isotopes is C-12 (98.93%) and C-13 (1.07%). If the abundance and mass of each isotope is known, the relative atomic mass of an element can be calculated by the formula:
isotope
Given the relative atomic mass of each element making up the molecule, relative molecular mass equal to the amount of relative atomic masses of all the atoms making up the molecule. Molecules that have a formula in one molecule AmBn means there tersbut m n atom atom A and B. Thus AmBndapat relative molecular mass is calculated as follows.
Mr-AmBn
10 Understanding And Personality Metal According Physicist
TOPICS: 10 Definition and properties of metals According Physicist
10 Understanding And Personality Metal According Physicist
DosenPendidikan.Com - In chemistry, a metal or metal categorized by Greek = Metallon is a chemical element that is ready to form ion cations and has metallic bonds, and sometimes it is said that it is similar to the cation in a cloud of electrons. Metal is one of the three groups of elements that can be distinguished by ionization and bonding properties, along with the metalloids and non-metals.
Metal
Metal
In the periodic table, a diagonal line drawn from boron (B) to polonium (Po) distinguishes metals from non-metals. Elements on this line are metalloids, sometimes called semi-metal, the element on the lower left is a metal, an element to the upper right is non-metallic.
The non-metallic in nature there are more than metal, but there are a lot of metal in the periodic table. Some well-known metals are aluminum, copper, gold, iron, lead, silver, titanium, uranium, and zinc.
Allotropes of metals tend to be shiny, soft, and a good conductor, while non-metals are usually brittle (for solid nonmetals), not shiny, and insulators.
In the field of astronomy, the term metal is always used to mention all the elements that memepunyai properties of objects heavier than helium.
Metal properties
chemical properties
Metals normally tend to form cations by removing electrons, then react with oxygen in the air to form an oxide bases. For example:
4 Na + O2 → 2 Na2O (sodium oxide)
2 Ca + O2 → 2 CaO (calcium oxide)
4 Al + 3 O2 → 2 Al2O3 (aluminum oxide)
Transition metals such as iron, copper, zinc, and nickel, takes longer to oxidize. Others, such as palladium, platinum and gold, do not react with the air at all. Some metals such as aluminum, magnesium, several kinds of steel, and titanium has a "patron" at the very outside, so are not able to be penetrated by oxygen molecules.
The process of painting, anodizing or plating on metal is usually the best steps to prevent corrosion.
physical properties
Electrical conductivity, thermal conductivity, luster and density properties. Metal that has a density, and hardness, and a low melting point example: metals generally have a high rate in the alkali metals and alkaline earth metals, are usually highly reactive. A high number of free electrons in any form solid metal causing the metal never look transparent.
The majority of the metal has a higher density than non-metals. However, variations in the density of this difference is very large, ranging from metallic lithium as the density of the smallest to osmium metal in the greatest density.
alloy
Alloy is a blending of two or more types of metal to get the physical, mechanical, electrical and better visual. Examples of metal alloys are popular is stainless steel which is a blending of iron (Fe) and chromium (Cr).
Use of Metal
Generally, the metal can be useful for people, for use in the field of industry, agriculture, and medicine. Such as the use of mercury in chlor alkali process. Chlor alkali process is an electrolytic process that plays an important role in manufacturing and purification chemicals.
Some of the chemicals that can be obtained by the process of electrolysis is sodium, calcium, magnesium, aluminum, copper, zinc, silver, hydrogen, chlorine, fluorine, sodium hydroxide, potassium bichromate and potassium permanganat.Proses electrolysis of sodium chloride solution is a process klor- alkali.
NaCl electrolysis to produce sodium hydroxide in the cathode (positive pole) and chlorine gas at the anode (negative pole). In the aerospace industry and the medical profession needed a strong material, resistant to corrosion, and noniritin, such as titanium alloys.
Most types of metal is an important element because it is needed in a variety of biochemical functions. In ancient times, certain metals, such as copper, iron, and tin used to make tools, machine tools, and weapons.
Precious metal
In general, precious metals mean metals, including alloys commonly used as jewelry, such as gold, silver, copper and platinum. These metals have a nice color, rust resistant, soft and present in small quantities in nature. Gold and silver have properties very good conductor of electricity so it is widely used to coat the connectors on an electronic device.
Heavy metal
Heavy metal (heavy metal) is a metal with a density of five or more, with an atomic number of 22 to 92. These heavy metals in the appraiser can harm health if it accumulates excessively in the body. Some are raised cancer (carcinogens). as is the case with foodstuffs with high heavy metal content deemed unfit for consumption.
The cases of environmental pollution causes a lot of foodstuffs containing excessive heavy metals. Case is a popular Minamata syndrome, as a result of the accumulation of mercury (Hg) in fish consumption.
In Indonesia, once proclaimed that the fish in the Bay of Jakarta also has a high mercury content. Shrimp from ponds in Sidoarjo been denied importers from Japan as it is considered to contain cadmium (Cd) and lead (Pb) which exceeds the threshold.
Allegedly these metals is the effect of the disposal of industrial waste around their environment. Cocoa from Indonesia was also rejected at an international auction because it is considered to contain cadmium above the permitted threshold. Cd probably derived from TSP fertilizer given to plants in plantations.





