TOPICS: Concepts Mol
Mol concepts
If in our daily lives we know the unit number in the form of a dozen (12 pieces), scores (20 pieces), and the rim (500 pieces) then the chemistry we know the mole as the unit number. One mole is defined as the amount of a substance containing particles according to the number of atoms in 12 grams of carbon-12. Since the mass of one atom C-12 is 1.992 × 10-23 grams, then the number of atoms in one mole (12 grams) of C-12 can be calculated as follows:
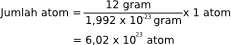
the number of atoms
Numbers 6.02 x 1023 is called Avogadro's number and Latau denoted by NA. Thus, one mole of a substance is the amount of substance containing 6.02 x 1023 particles. If n states the number of moles and N denotes the number of atoms or molecules, then the relationship n and N can be written in the following formula.
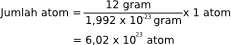
Molar mass
The molar mass is the mass of one mole of a substance. The value of the molar mass of a substance is always the same with Ar or Mr substances. The relationship of mass and number of moles of a substance can be written in the form of the following formula.
gram-mole
Volume Molar Gas
Gas molar volume is the volume of 1 mol of gas. Due to the volume of gas is strongly influenced by temperature and pressure, the stoichiometry of the chemists define a reference condition in the determination of the molar volume. This reference condition is 0 ° C (273 K) and 1 atm. This condition is called standard conditions or STP (Standard Temperature and Pressure). At STP conditions, molar volume of gas is 22.4 L. Thus, the relationship number of moles of gas (n) with its volume can be written by the following equation.
volume-mol
Volume (V) and the molar volume (Vm) gas expressed in liters.
- See more at: http://apapikir.com/2016/02/12/konsep-mol/#sthash.AxJOz9xS.dpuf

0 comments:
Post a Comment