This is default featured slide 1 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 2 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 3 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 4 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
This is default featured slide 5 title
Go to Blogger edit html and find these sentences.Now replace these sentences with your own descriptions.This theme is Bloggerized by Lasantha Bandara - Premiumbloggertemplates.com.
Saturday, 13 February 2016
Branches of Chemistry together with the insight
Dalton's Atomic Theory
The reaction equated with Method Ping pong
Mol concepts
TOPICS: Concepts Mol
Mol concepts
If in our daily lives we know the unit number in the form of a dozen (12 pieces), scores (20 pieces), and the rim (500 pieces) then the chemistry we know the mole as the unit number. One mole is defined as the amount of a substance containing particles according to the number of atoms in 12 grams of carbon-12. Since the mass of one atom C-12 is 1.992 × 10-23 grams, then the number of atoms in one mole (12 grams) of C-12 can be calculated as follows:
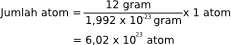
the number of atoms
Numbers 6.02 x 1023 is called Avogadro's number and Latau denoted by NA. Thus, one mole of a substance is the amount of substance containing 6.02 x 1023 particles. If n states the number of moles and N denotes the number of atoms or molecules, then the relationship n and N can be written in the following formula.
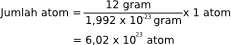
Molar mass
The molar mass is the mass of one mole of a substance. The value of the molar mass of a substance is always the same with Ar or Mr substances. The relationship of mass and number of moles of a substance can be written in the form of the following formula.
gram-mole
Volume Molar Gas
Gas molar volume is the volume of 1 mol of gas. Due to the volume of gas is strongly influenced by temperature and pressure, the stoichiometry of the chemists define a reference condition in the determination of the molar volume. This reference condition is 0 ° C (273 K) and 1 atm. This condition is called standard conditions or STP (Standard Temperature and Pressure). At STP conditions, molar volume of gas is 22.4 L. Thus, the relationship number of moles of gas (n) with its volume can be written by the following equation.
volume-mol
Volume (V) and the molar volume (Vm) gas expressed in liters.
- See more at: http://apapikir.com/2016/02/12/konsep-mol/#sthash.AxJOz9xS.dpuf
The relative atomic mass and molecular mass Relative
TOPICS: relative atomic mass and molecular mass Relative
The relative atomic mass and molecular mass Relative
Source: http: //www.mystupidtheory.com/
Atoms are very small particles. Because the atomic mass is also too small when expressed in units of grams so that the chemists created a way to measure the mass of an atom, namely the relative atomic mass. The relative atomic mass (Ar) is the ratio of the average mass of an atom with one-twelfth times the mass of one atom of carbon-12.
Ar
The smallest unit of a substance can also be a molecule. Molecules composed by two or more atoms held together by chemical bonds. Relative molecular mass (Mr) adalahperbandingan average mass of a molecule with one-twelfth times the mass of one atom of carbon-12.
Mr
In the above formulas used atomic mass and molecular mass on average. Why use an average atomic mass? Because the elements in nature have beberapaisotop. For example, the carbon in nature has two pieces of stable isotopes is C-12 (98.93%) and C-13 (1.07%). If the abundance and mass of each isotope is known, the relative atomic mass of an element can be calculated by the formula:
isotope
Given the relative atomic mass of each element making up the molecule, relative molecular mass equal to the amount of relative atomic masses of all the atoms making up the molecule. Molecules that have a formula in one molecule AmBn means there tersbut m n atom atom A and B. Thus AmBndapat relative molecular mass is calculated as follows.
Mr-AmBn
10 Understanding And Personality Metal According Physicist
TOPICS: 10 Definition and properties of metals According Physicist
10 Understanding And Personality Metal According Physicist
DosenPendidikan.Com - In chemistry, a metal or metal categorized by Greek = Metallon is a chemical element that is ready to form ion cations and has metallic bonds, and sometimes it is said that it is similar to the cation in a cloud of electrons. Metal is one of the three groups of elements that can be distinguished by ionization and bonding properties, along with the metalloids and non-metals.
Metal
Metal
In the periodic table, a diagonal line drawn from boron (B) to polonium (Po) distinguishes metals from non-metals. Elements on this line are metalloids, sometimes called semi-metal, the element on the lower left is a metal, an element to the upper right is non-metallic.
The non-metallic in nature there are more than metal, but there are a lot of metal in the periodic table. Some well-known metals are aluminum, copper, gold, iron, lead, silver, titanium, uranium, and zinc.
Allotropes of metals tend to be shiny, soft, and a good conductor, while non-metals are usually brittle (for solid nonmetals), not shiny, and insulators.
In the field of astronomy, the term metal is always used to mention all the elements that memepunyai properties of objects heavier than helium.
Metal properties
chemical properties
Metals normally tend to form cations by removing electrons, then react with oxygen in the air to form an oxide bases. For example:
4 Na + O2 → 2 Na2O (sodium oxide)
2 Ca + O2 → 2 CaO (calcium oxide)
4 Al + 3 O2 → 2 Al2O3 (aluminum oxide)
Transition metals such as iron, copper, zinc, and nickel, takes longer to oxidize. Others, such as palladium, platinum and gold, do not react with the air at all. Some metals such as aluminum, magnesium, several kinds of steel, and titanium has a "patron" at the very outside, so are not able to be penetrated by oxygen molecules.
The process of painting, anodizing or plating on metal is usually the best steps to prevent corrosion.
physical properties
Electrical conductivity, thermal conductivity, luster and density properties. Metal that has a density, and hardness, and a low melting point example: metals generally have a high rate in the alkali metals and alkaline earth metals, are usually highly reactive. A high number of free electrons in any form solid metal causing the metal never look transparent.
The majority of the metal has a higher density than non-metals. However, variations in the density of this difference is very large, ranging from metallic lithium as the density of the smallest to osmium metal in the greatest density.
alloy
Alloy is a blending of two or more types of metal to get the physical, mechanical, electrical and better visual. Examples of metal alloys are popular is stainless steel which is a blending of iron (Fe) and chromium (Cr).
Use of Metal
Generally, the metal can be useful for people, for use in the field of industry, agriculture, and medicine. Such as the use of mercury in chlor alkali process. Chlor alkali process is an electrolytic process that plays an important role in manufacturing and purification chemicals.
Some of the chemicals that can be obtained by the process of electrolysis is sodium, calcium, magnesium, aluminum, copper, zinc, silver, hydrogen, chlorine, fluorine, sodium hydroxide, potassium bichromate and potassium permanganat.Proses electrolysis of sodium chloride solution is a process klor- alkali.
NaCl electrolysis to produce sodium hydroxide in the cathode (positive pole) and chlorine gas at the anode (negative pole). In the aerospace industry and the medical profession needed a strong material, resistant to corrosion, and noniritin, such as titanium alloys.
Most types of metal is an important element because it is needed in a variety of biochemical functions. In ancient times, certain metals, such as copper, iron, and tin used to make tools, machine tools, and weapons.
Precious metal
In general, precious metals mean metals, including alloys commonly used as jewelry, such as gold, silver, copper and platinum. These metals have a nice color, rust resistant, soft and present in small quantities in nature. Gold and silver have properties very good conductor of electricity so it is widely used to coat the connectors on an electronic device.
Heavy metal
Heavy metal (heavy metal) is a metal with a density of five or more, with an atomic number of 22 to 92. These heavy metals in the appraiser can harm health if it accumulates excessively in the body. Some are raised cancer (carcinogens). as is the case with foodstuffs with high heavy metal content deemed unfit for consumption.
The cases of environmental pollution causes a lot of foodstuffs containing excessive heavy metals. Case is a popular Minamata syndrome, as a result of the accumulation of mercury (Hg) in fish consumption.
In Indonesia, once proclaimed that the fish in the Bay of Jakarta also has a high mercury content. Shrimp from ponds in Sidoarjo been denied importers from Japan as it is considered to contain cadmium (Cd) and lead (Pb) which exceeds the threshold.
Allegedly these metals is the effect of the disposal of industrial waste around their environment. Cocoa from Indonesia was also rejected at an international auction because it is considered to contain cadmium above the permitted threshold. Cd probably derived from TSP fertilizer given to plants in plantations.
Friday, 12 February 2016
Types of Synthetic Sugar and health impacts
Chemicals in Laboratory Glassware well as type-kind [Complete infographic]
TOPICS: Glass Chemistry Laboratory And Type In-Kind [Complete infographic]
POSTED BY: NANDASANJAYA FEBRUARY 12, 2016
Chemicals in Laboratory Glassware well as type-kind [Complete infographic]
Source: http: //www.mystupidtheory.com/
One thing that is very important in the laboratory is the use of various types of beaker. Beaker has many forms, and each form has a goal and their respective functions. During the conduct of research in chemistry lab, you will be in direct contact with various forms beaker. Therefore, knowledge about it is very important to you.
Development of various types of chemical glassware has been done since the time of Jabir Ibn Hayyan, the founder and the foundations were chemistry. Somehow I make and what technology is used, but at the time it was created several beaker for the purposes of the experiment and calculation.
equipment beaker
equipment beaker
So much for the importance of the role of the beaker in an experiment, then as a student of science, you have to recognize any kind of this beaker.
Basic Chemistry glass
Equipment beaker is definitely there in every Laboratory of Chemistry is Test Tube (test tube), tube Boiling (boiling tube), and a glass beaker (beaker glass). In Indonesia Setau I, Boiling Tube is usually equated with the Test Tube. Actually, the difference is slim, if the test tube ends mediocre, but if the boiling tubes at the edges there are grooves, these grooves serves to remove the heat on the tube so it can be held. Have a look at the picture:
beaker base
beaker base
Retort
Some kind of retort which is common in the laboratory is Pumpkin Erlemayer (Canonical Flask), Tubes Volumetric (volumetric flask), Pumpkin Alas Round (round bottom flask), Pumpkin Florence (florence flask), Pumpkin Kjehdahl (Kjehdahl Flask), Pumpkin Pear (pear shaped flask), pumpkin retorsi (retort flask, Schlenk flask (Schlenk flask), pumpkin straus (straus flask), pumpkin Claisen (Claisen flask).
beaker tube
tube beaker
Of all the chemical tube mentioned above, the most famous being flask which was discovered by German chemist named Emil Erlenmeyer. Volumetric flask will very often encountered when manufacturing of standard solutions and preparation of the solution, because it is used in determining the standard volume of solution.
Some functions and the use of chemical canisters would I describe in the next post.
Funnel and glass dividers
Some glassware is very important, especially when working in a chemical laboratory and separating chemical solution. Glassware this funnel of which is Funnels Filter (filter funnel), Funnels Thistle (thistle funnel), tube Buchner (Buchner funnel), funnel separator (separating funnel), Funnel Drops (Dropping Funnel), Column Chromatography (Chromatography Column) and Tubes Thiele (Thiele tube).
glass separating funnel
glass separating funnel
Among all those presented above, I'd never seen in Laboraturium just Thiele tube. Sealin it, are all important glasses are indispensable in research in the field of chemistry.
condenser Heat
The condenser is a chemical laboratory equipment that is essential in experiments, particularly in the field of organic chemistry. Some types of condenser glassware in the laboratory is Liebig Condensers, Graham Condenser, Condenser Friedrichs and fractionation columns.
In general, the use of the condenser is to cool the steam, so that steam will be used in the separation of liquids and a solution.
glass condenser
glass condenser
Measuring cups
Beaker commonly used in the measurement of which is Tubes Measure (graduated cylinder), Pipette Volume (volumetric pipete), Pipette Measure (graduated pipete), Buret (burette), and Syringe Gas (gas Syringe).
chemical measuring cup
chemical measuring cup
These measuring cups are used in experiments that require precision measurement, ie quantitative analysis. Each type of glass used for different purposes in accordance with the functions and accuracy of every instrument.
source picture and material: http://www.compoundchem.com/2015/03/17/glassware/
10 Characteristics of Chemical Reaction Most Easily Observed
TOPICS: 10 Characteristics The Most Easy Chemical Reactions Observed
10 Characteristics of Chemical Reaction Most Easily Observed
Source: http: //www.mystupidtheory.com/
One of the discussion of important chemical reactions is kimia.Reaksi Chemistry is the process of change of a chemical compound (reactants) into other chemical compounds (products). When there is a chemical reaction, we will never get to see the inception of a new compound by naked eye.

The characteristics of chemical reactions
But in this process are the characteristics of a chemical reaction has occurred. There are ten different signs that characterize the chemical change in a substance:
The characteristics of occurrence of Chemical Reactions
1. The formation of gas bubbles.
Gas produced in a chemical reaction sometimes causes the formation of bubbles.
Bubbles can arise from heating a liquid or gas formation in the liquid. Both of these events is a common chemical reactions occur.
2. Establishment Deposition.
When mixing the two substances that are soluble in the solvent, then formed solids at the bottom of the solution, indicating the occurrence of chemical reactions in the solution.
The formation of this precipitate sometimes in the form of very small particles (micro) floating in solution so it is not visible. To make sure you can meyenterkan light on the solution. This phenomenon is known as the Securities Brown (Brown Effect)
3. The occurrence of Color Changes.
Some chemical compounds have a tendency to absorb and emit colors. The ability to absorb and emit very specific color for some substances. So when there is a change (conversion) substances of reactants into products (new compound) there will be a color change.
It could be from two reactants colorless will produce colored compounds, or of colored reactants to form a colorless product. This incident shows the change of chemical (Chemical Reaction).
4. Temperature Change.
Every chemical compound has the energy. This energy is the energy in the chemical bonds within a compound. Break the bond requires energy, while the formation of chemical bonds release energy.
When the chemical reactions that occur melibatakan more than the formation of chemical bonds breaking chemical bonds, it would be excessive and the energy is released as heat and the temperature rises.
However, if a chemical reaction involving more break the bond than the bond formation, it will be less and the energy absorbed from the environment led to the cold and decreasing temperature.
5. Transmitting light.
Chemical reactions occasionally releases energy in the form of light. Examples of this chemical reaction is that the fireflies emit light when night.
6. Volume Changes occurred.
When a new compound (product) is formed, then the reactants compound will be reduced drastically. The formation of new compounds and the reduction of these reactants can cause drastic changes in density so that the volume change.
7. Changes in conductivity.
Some chemical reactions produce or modify ion daapat into other compounds. This will affect the change in conductivity of a system.
8. There is a change Bau.
Some chemical compounds (such as ammonia) have strong odors. Hence the formation of ammonia or other smelling compounds will cause changes in odor on the system.
9. Changes in Taste.
Chemical compounds that have a particular flavor will result in change in taste when it reacts. This can be easily observed when we boil the potatoes, initially not too sweet potato flavor, then boiled glucose will be formed which has a sweet taste.
10. There is a change Boiling and Freezing Point.
Generally this happens in the crystal. To find a crystal has reacted or not, can use the boiling point and freezing point.
That's 10 Characteristics of chemical reactions that have occurred in a system.
Physics as Science Basis
TOPICS: Physics For Basic Sciences
Physics as Science Basis
Basic Sciences, Physical Education

Laser, one technology that uses theoretical basis of quantum physics
Nowadays humans so is facilitated by a wide range of existing technologies. The technology in the field of transport allows humans to travel far in a reasonable time. Aircraft, ships, trains, buses, cars, motorcycles, and bicycles are several means of transport that have been created by people. with their means of transport is, people can travel from one place to another and connect with people in many other places on earth. All of this transport technology will not be possible to develop and create without the foundation of science that supports it. The cornerstone of science for all this technology is none other than the natural sciences and especially physics and mathematics.
Likewise in the field of communications and computers, has now developed specific areas in the field of communications and computer called information and communication technologies (ICTs) or in terms of the English information and communication technology (ICT), technological progress has taken place so rapidly. With communication technology, people can now communicate and interact even in a very long distances. With the telephone has evolved into a phone (mobile phone), people can talk and interact from a distance and without the need to meet in person.
Especially with the advent of computers and the Internet, so many conveniences that can be enjoyed by people who previously could not and I never thought to do it. All of these technologies can be created naturally in line with the development of scientific theories, principles and underlying concepts.
Airplanes and rockets can be made based on hukumNewtondan Bernouli. Ships and submarines with regard to natural law propounded by Archimedes. Computers in the form of a simple and compact can be made after the creation of transistors and ICs that utilize semiconductor material whose theory proposed by some quantum physicists. Lasers are widely used in the CD player and some medical devices utilizing quantum physics theory that has been put forward by Einstein, Pauli, Heisenberg, and comrades. And, there are many new tools and technologies that can be designed and manufactured using the existing physical theories.
No exaggeration if we say that the physics is a basic science or basic science of science and technology there. Concepts and principles in physics are widely used to build or establish new technologies. Scientific physics itself continues to evolve to be able to find an explanation or a new theory that could explain the new phenomena. In fact, this time the physical sciences are also beginning to be used in social and economic fields. Principles and theories in physics began to be used to find solutions or explain the phenomenon in the social and economic.
Physics and mathematics are widely used in the manufacture of new technologies for physics and mathematics has provided the theoretical basis and scientific background. This physical principle is then shaped in the form of a mathematical model in the form of a mathematical equation for a particular problem which will then be sought after solution of the equation. This applies in all fields including engineering and also social and economic.
Physics also provides scientific and reasonable explanation of an event of natural or technical factors that need a reason or explanation. For example, an engineer who wants to design a bridge would have to take into account everything that made the bridge will be strong and resistant to physical disorders. The calculation of the course covers all the theories and concepts of physics that apply to that bridge and using mathematical models as appropriate. Theoretical physics will always be used in the creation and establishment of new technologies.
This is why physics with mathematics known as basic science. Principles, theories, and concepts used in the field of science and technology there.
Benefits Life and Learning them
TOPICS: Benefit Life And Learning them
POSTED BY: NANDASANJAYA FEBRUARY 11, 2016
Benefits Life and Learning them
benefits Biology
Benefits Biologi- This time it will be explained about the benefits of biological sciences, biology and benefiting the benefits of studying the biology of various spheres of life such as industry, agriculture, medicine, pharmacy. Let belajarbiologi.
Benefits biology | With the development of technology and other sciences that led to the development of biological sciences and knowledge of our living beings as well as an increase in the understanding of the surrounding environment, biology provides many benefits as well as contributions to the progress of society. The biological benefits not only the benefits but the benefits of such a study would be a more realistic and applicable of course, especially in various fields such as agriculture, animal husbandry, medicine, industry, healthcare and pharmaceuticals. Here's a brief description of the biological effect in our lives, especially in various fields areas of society.
Benefits of Biology in Agriculture
Benefits of biology in agriculture - The population is increasing. This should be accompanied by an increase in food production. Biology has contributed to efforts to increase food production. Benefits of biology in agriculture is certainly much needed due to population growth is fast enough so it must be balanced with food production were traveling too fast to avoid starvation and increased high prices due to scarcity of goods. Some examples of the use of biology in the field of agriculture are as follows:
Some of the previous century farmers are planting both traditional agriculture and plantation. But with the development of knowledge about seeds, fertilizing patterns and the use of organic pesticides, farmers were able to increase the amount of production for each area of the existing plant area. Even speed up the time of harvest that were once only able to harvest 1-2 times per year, has now been 3 times for each year. Of course, this is a benefit of the combination of biology and other sciences, especially of agricultural science.
Benefits of biology in agriculture one of which again is genetically engineered. Through the development of biotechnology and molecular biology, biologists have successfully developed genetically rekaya to plant particular crops consumed by humans. The application that has been successfully carried out is the creation of cultivated plants that are able to produce its own insecticide, so that the plant does not need to be sprayed with insecticide. Examples of crops that have been successfully engineered with these objectives are fruit trees, apples, pears, kale / cabbage, broccoli, and potatoes. With the existence of genetic engineering, there was indeed a loss in it, namely the loss of germplasm due knocked out of the people's choice, however, one major problem: the need for food can be met.
In addition, the biological effect is no less important in the field of agriculture is the tissue culture technique. This technique menggadakan plants in a short time and practical so as to increase seed production and propagation. By using one tanamaan superior results and genetically healthy and able to produce millions and millions of plants of same nature so that agriculture will be assured the results are able to provide good for society and economies. It also shortens the time in nursery plants grown due to already form a complete plant, staying grown for harvest.
In addition, no less important benefit is the understanding of the genetics of living beings make farming more advanced. New crop varieties that are superior and more superior than the previous crop can be produced by the science of genetics. How? You need to know the benefits of biology in agriculture described above is still the big picture, there are many other biological benefits in the agricultural sector has been well applied up to now.
The biological effect in the livestock sector
Benefits of biology in the field of animal husbandry - One of the things that are important as a source of food and nutrition / human nutrition is livestock. In connection with the increase in population and the need for nutritious food and protein, biological science knowledge is needed benefits in this field, in particular section livestock production and genetically modified (Although it is still forbidden needed but has done a lot of research for this). Here are some of the biological effect in the livestock sector:
In biology is divided into several specialization of science and some parts of the role in the field of animal husbandry such as anatomy, physiology of animal genetics and animal embryology. By studying some of the science, now it has been applied on a large a large so-called artificial insemination. Artificial insemination is a method used by farmers to increase the number of livestock owned by females fertilize using a syringe prefilled male sperm. With this, the female can become pregnant cattle without the need for the marriage process are usually so as to accelerate the pregnancy cow, and we can choose to set a male superior to the cow.
Besides artificial insemination techniques, today has developed the technique of in vitro fertilization.
In addition, the manufacture of silage which is a food or feed cattle and other livestock are produced from the fermentation of plant or other material that is high cellulase thereby increasing its protein content that makes a cow a cow and cattle weights were able to rise rapidly.
With the discovery of vaccines and antibodies as well as prebiotics, animal health can be improved
In addition, the biological effect in the livestock sector is still under development is at the genetics and molecular biology. Various farm animals that have been genetically engineered either directly or indirectly (using a system of crossbreeding) to find a race of races that excel in a particular field, such as for broilers specifically for meat only, then in addition to use drugs and hormones, is also used race chicken which is much faster in the growth process the meat.
Benefits biology in medicine
Biology provides a great benefit to the advancement of medical science, especially from administration of the disease problems. Here are a few examples of the benefits of biology in medicine:
Transplants (transplants) organ has to do in medical science. Although indeed these benefits not only the biological effect alone but a combination of some of the science, but by studying biology in particular of human anatomy, animal anatomy and physiology of animals, we are able to solve the mystery of replacement organs that have been damaged, and knowledge about the rejection of organ which did not match (match) can we press uses the immune system suppressing drugs.
Virology is one branch of biology who specializes in studying and researching the virus, a transitional living creatures (although there are some experts who regard as living beings). Virology plays an important role in various diseases suffered by humans for example, just the other is the name of the H5N1 bird flu virus. To find the vaccine necessary knowledge about this virus that the doctors can perform precise handling.
IVF techniques. In the biological sciences, especially embryology, described the animal and human embryos. Such knowledge combined with a lot of trial and experimentation have produced test tube baby technique. The biological effect that this one makes the couples who have difficulty conceiving or even a particular case wants to get pregnant but sterile can have children. With the tenik IVF child's gender can be determined up to 90% since before conception, sperm and ovum will perform a fusion selected from chromosomes, and more on genetic conditions. After forming the zygote (still in vitro), at the morula stage, the embryo will be inserted into the mother's uterus, or womb of a foster mother to be a perfect baby.
Medical microbiology. Microbiological knowledge that develops very rapidly to make the benefits of biological increasingly felt. Benefits of biology in medicine at the microbiological treatment of disease is a disease caused by bacteria and other micro organisms that are more effective, and the use of antibiotics is the result or product of microorganisms in general.
Benefits of biology in the field of industry
You must know biotechnology. In industry, the biological sciences have indeed tasted especially the name of biotechnology. The use of living organisms in the industry open up new land for workers, self-employed and more and improve existing food. For example only, the dairy industry is using the technique of pasteurized fresh milk from cows that can be taken and still kept clean even after long storage (still closed). Then the yoghurt industry that uses bacteria and fermentation techniques in manufacturing, industrial soy sauce, candied industry and many other industries are now using biotechnology due to a more friendly, economical and healthy for consumers.
Section of the health industry, drugs (Pharmaceutical) and in particular antibiotics, biology is very helpful. Especially the first time it was found that microorganisms capable of producing anti-life or compounds capable of killing the organisms around it that is now called antibiotics. With this knowledge created penicillin, and many other drugs produced from microorganisms. And even vitamin vitamins that often there to drink, some products on the market developed from products of microorganisms.
the biological effect and the benefits of studying biology
benefits Biology
And what about the benefits of studying biology
Parts And Functions BakteriBelajar Bacterial Cells:
TOPICS: Parts And Functions BakteriBelajar Bacterial Cells: StrukturPengertian
POSTED BY: NANDASANJAYA FEBRUARY 11, 2016
Studying Bacteria Cells: Structure Definition, Parts and Functions Bacteria
Source: http: //belajarbiologi.com/
Learning Biology: Cell Bacteria
Bacteria is one of the living beings. Domain bacterial cells are in prokaryotes which means it has no nuclear membrane on its cell nucleus so that the core or nucleus of the bacterial cell spread. The nucleus of cells and the spread of bacteria called nukleoida (clear areas containing DNA in bacteria). One example of a bacterial cell is Escherichia coli, Bacillus subtilis, Salmonella typhimurium.
Structure of Bacterial Cells
Bacteria are single-celled, or unicellular creatures that do not have a nucleus wrapping. Apart from that, bacterial cells do not contain organelles organelles that are often found on eukaryotic cells such as animal cells and plant cells.
Before explaining more about the structure of the bacterial cell, you need to learn about many different forms of bacterial cells or bacterial cell morphology.
Here's an explanation of sorts forms of bacterial cells
Miscellaneous forms of bacterial cells
Broadly speaking, the form of bacterial cells composed of four kinds:
cocci
bacillus
Spiral
filamentous
Cocci bacteria cells are shaped like a ball-shaped or circular (2D). Bacterial cells that form spherical cocci or have some kind of direction that affects the formation of colonies of bacteria that form monokokus, diplokokus, diplococci, staphylokokus, Streptococcus, sarkina and tetrakokus.
Monokokus bacteria cells have a single form or attaches between one cell to another cell. Diplokokus bacterial cells divide by two two-cell or two cells each attaches. whereas staphylokokus bacteria have cell division in all directions except to the bottom and to the top of the field shaped bunches of grapes, while Streptococcus splitting lengthwise into two directions over 2 sarkina bacterial cells and cells divide like a cube 8-cell or 4-cell and 4-cell above the bottom. While tetrakokus bacterial cells divide every four cells in all directions except the top and bottom.
Studying Bacteria Cells: Structure Definition, Parts and Functions Bacteria
Cocci bacteria cell shape
Shaped bacillus bacteria cells have some form of division or form colonies like cocci-bacilli, monobasil, diplobasil, palisades (fences), and streptobasil. Shape cocci bacilli in bacterial cells means elliptical, shape monobasil bacterial cells means consisting of a single cell and attaches to other cells when to divide, while diplobasil splitting with two two cells. Palisades form of bacterial cells divide like a fence or cut laterally so that the bacterial cell is seen standing side by side with pembelahannya bacterial cells, while the shape of bacterial cells divide streptobasil like elongated chain.
Studying Bacteria Cells: Structure Definition, Parts and Functions Bacteria
Bacillus bacterial cell shape: Various kinds of bacterial cells
Spiral-shaped bacterial cell you can imagine as you twist the fabric. Spiral-shaped bacteria cells there are several kinds of spiral berdiperbesar, vibrio, a comma-shaped, spiral mace, and helical spiral. There is also a bacterial cell that is shaped like Borrelia burgdorferi bolts.
Bacterial cell filaments shaped like a mushroom shape or cells of hyphae in length. Besides other bacterial cell filament-winding is a spirochete.
Bacterial form spiral: Various kinds of bacterial cells
Bacterial form spiral: Various kinds of bacterial cells
After this let's learn the structure of the bacterial cell and the section parts
Bacterial cell structure and Section Section Bacterial Cells
The cell structure of bacteria in general everything is almost the same shaped nukleous nukleoida. the cell walls of bacteria, as well as minor form of plasmids and chromosomes contained or pili and pilus ribosomes. plasma membrane, and mesosom. Other extras like flagellum (flagella), S layer, karboksisome, magnetosome can be found in certain bacteria.
Here's a picture bacterial cell structure:
Studying Bacteria Cells: Structure Definition, Parts and Functions Bacteria
Structure of Bacterial Cells
Let's discuss the start of the bacterial cell wall. Bacterial cell walls are composed of peptidoglycan. Peptidoglycan is a polysaccharide chain composed of acid N-Asetilmuramik (NAM) and N-acetylglucosamine (NAG). Bacterial cell wall function in maintaining cell shape, maintain turgor pressure in the cell or cells due to the rich content of proteins in a cell. Cell wall in bacteria are divided into two types of gram positive and gram negative bacteria. Difference between the two lies in the amount of peptidoglycan and the specific characteristics of the membrane cytoplasm.
Plasma membrane of bacteria or cytoplasmic membrane, generally composed of a phospholipid bilayer. However, specific to gram-negative bacteria, has the outer membrane and periplasmic before reaching the cytoplasmic membrane so it looks like three layers. Layer outer membrane composed of lipopolysaccharide and protein. It distinguishes between gram-negative and gram-positive bacteria.
Studying Bacteria Cells: Structure Definition, Parts and Functions Bacteria
The cell wall structure of bacteria
Studying Bacteria Cells: Structure Definition, Parts and Functions Bacteria
Layer of the bacterial cell membrane
Flagella or flagellum (single) in bacterial cells function as locomotor bacteria. There are several kinds of flagella in bacteria based on the location and the amount in the bacterial cell. For more details about the bacterial cell division by flagellanya please read this article on the motility of bacteria.
Fimbriae or hair cells is a protein tubes that are on the outside of the cell. Its main function in attachment fimbriae but in some cases such as Myxococcus can function in motility of bacteria to form fruit bodies.
Besides fimbriae, there is also pili, has the same structure as fimbriae but longer and fewer of the bacteria. This is because the functions are more specific to conjugation as well as strengthen the adhesion of bacteria.
The inside of the cell are the ribosomes of bacteria that play a role in protein synthesis. Ribosomes in bacterial cells in contrast to eukaryotic cells in terms of size. The size of the bacterial ribosome is 70 S (S = Svedberg), with a large subunit and a small subunit 50 S 30 S.
In addition there are also ribosome cytoskeleton. Cytoskeleton function of the bacterial cell together with the eukaryotic cytoskeleton.
There is also the inclusion in bacterial cells such as fat droplets, crystals, glycogen, and pigments. Inclusion of bacterial cell has no membrane and does not metabolize.
In addition, there is also a gas vacuole. However vacuole gas only found in certain bacteria such as planktonic bacteria and cyanobacteria.
Bacterial cell also has a circular DNA and in some bacteria are linear DNA. Additionally, bacteria also have a DNA plasmid or mini functioning in forwarding certain information. Often referred to as extrachromosomal plasmid DNA.
Mesosome formerly known as a folding membrane produced by bacterial cells as an intracellular organelle or membrane, it was found as an artifact caused by the process of doing the observation using an electron microscope. However, there are several kinds of bacteria as intracellular membranes in bacteria fototrofik chromatophore, containing bakterioklorofil and carotenoids.
In addition there is also mikrokompartemen in bacteria such as karboksisome and magnetosome. Mikrokompartemen function in the metabolism of certain specific bacteria and wrapped so that it does not contain lipid protein because it is not covered by a membrane.
It seems like almost all the parts and structures of bacteria you have learned. For more details, I suggest you read a book specifically on bacteria such as bacteriology books.
Let us make sense of the bacterial cell so clearly defined. Definition of a bacterial cell is an organism that has the characteristics of living things with the structure of the cell walls are composed of peptidoglycan and has a very small size and does not have a nuclear membrane so have nukeleoida and are unicellular.
So and studying biology with the spirit yah ...
Bacteria Cells Keyword:
Cells, bacteria, Peptidoglycan, cell walls, organelles, ribosomes, Karboksisome, magnetosome, cell membrane, the cell structure of bacteria, the bacterial cell membrane, the cell wall of bacteria.
Definition, structure and function of cells and organelles
TOPICS: PengertianStruktur And Function And Organelles Cells
POSTED BY: NANDASANJAYA FEBRUARY 11, 2016
Definition, structure and function of cells and organelles
Source: http: //belajarbiologi.com/
Understanding cell | Cell Structure and Function | The cell organelles | This time belajarbiologi.commenjawab of your questions about the understanding of the cell, how the structure of the cell, how cells function and function of organelles in the cell organelles of cells and many kinds of cells that existed until now. Let's study the cell biology.
Understanding Cell and Cell Theory
What is the cell? In biology, cell material is studied in cell biology is called. Own cells was discovered accidentally by Robert Hooke although by this time he simply observed the dead cells from the tissue of cork. Therefore, he called cella or empty space is then in English called the cell which then absorbed into Indonesian as cells. Can you realize that the word cell, may seem a bad analogy for a living creature's smallest functional. Just imagine, the room is empty, even the room where the arrested criminal is often referred to as a holding cell. Well, but that's the reason, because Robert Hooke only observed an empty cell death and to honor his discovery, so until now researchers have named cells. So what is a cell? Understanding mashyur cells most known is from the duo researchers that Schleiden and Schwann cells are on the theory that a combination of several previous studies by researchers bebeberapa of cells such as Robert Virchow, H.J. Dut Rochet, Max Schultze, Robert Brown, Antony van Leeuwenhoek. There are three theories duo cells of these researchers are:
All living organisms are composed of cells or cells. An important definition of a cell is that it is a unit of protoplasm surrounded by a thin plasma membrane. The protoplasm of all cells Contain a nucleus. The rest of the protoplasm is called cytoplasm or cytosol. The cytoplasm contains many cell organelles. This means that all living things are composed of either a single cell (unicellular) or many (multicellular). Understanding cell main thing is that each cell is a unit of protoplasm enveloped by plasma membrane (thin membrane). Protoplasm in all living cells contain a nucleus or the cell nucleus. Aside from the cell nucleus, cytoplasm contained in the cell or can be called cytosol. In the cytoplasm of cells, there are some cell organelles.
Cell are the functional unit of life. That is, the cells are functional units in living things. Every living creature in this world until now arrayed by the cell or cells so that it can carry out character traits as living beings. Cells in multicellular organisms interact with each other and work together to form tissues and then form organs and so on up into the individual unit of living things. Whereas in single cells, organelles of the cell organelles carry out its functions efficiently and controlled by the cell nucleus (nucleus) in order to carry out the character as living beings (described in more detail later) and every kind of unicellular and not even able to do the kind of communication and interaction
Each Arise cells from pre-existing cells. That is, each cell that has been present there due to the cells forming. The above sentence describes the cells' ability to multiply or divide either sexually or asexually paraseksual. This capability makes the existing cells capable of sustainable. It still supports the theory that modern abiogenesis their protobion before then presenting cells such as cells that you see today. The above statement does not justify the theory of biogenesis, so you do not misunderstand that the three above cell theory states that all cell types have been there before. Not at all, the theory of evolution to justify the above cell and directs the cell to the cell's ability to reproduce themselves. In addition, the third cell theory also explains about the ability of certain cells to perform a fusion or merger as in the egg and sperm to form a zygote, and the ability of the cells to other cells such as diploid cell into four haploid cells in spermatogenesis.
Based on the theory of the cell by Schwann and Schleiden above, it can be concluded that the definition of a cell is
The cell is the functional unit of life that is a constituent of living beings or living beings composed of protoplasm surrounded by a thin membrane and is able to reproduce itself either sexually or otherwise so as to form daughter cells either identical or not.
Based on the understanding of cells above, it is clear that some of the findings today such as viruses and prions his comrades also not a living thing because not fit with the notion of cells above. If the virus is said to be an absolute cell already, the virus is a living creature. So, you do not say "cell virus" Well, enough virus alone.
Various kinds of cells (Brief)
Before more in discussing about the cell, the cell structure and cell organelles, let us know briefly kinds of cells that exist today. Broadly speaking, the cell is divided into two based on the presence or absence of nuclear membrane is a eukaryotic cell (having nuclear membrane) and prokaryotes (does not have a nuclear membrane). This is broadly split all branches of living things that we know today. Starting from archaebakteria, bacteria and eubacteria (all living organisms other than bacteria and archaebacteria).
Furthermore, cells divided more specifically into cells unicellular animals (protozoa), unicellular algae cells, cell fungi (hyphae), plant cells, multicellular animal cells, bacterial cells, cell archaebakteria, and various types of cell differentiation that exists. Overall there are cells that are all adapted to their habitat are and kebutuhakan them to remain stable.
Definition and Structure of Cells
In this article we'll describe 4 + 1 kind of cell structure that exists is the structure of multicellular animal cells, plant cell structure, cell structure unicellular animals (protozoa), the cell structure of bacteria, and the last structure of unicellular algae cells.
Understanding the structure of the cells of multicellular animals
You know what that make up your body? It is animal cells together to form tissues, and organs and organ systems and be human. Understanding animal cells are cells that have the protoplasm and the cell membrane but does not have a cell wall as well as the ability to photosynthesize, besides understanding animal cell is a cell that is heterotrophic organisms and does not have a cell wall.
The structure of animal cells are generally composed of cell membranes and protoplasm. Animal cell protoplasm there is cytoplasm and nucleus of the cell (nucleus) as well as cell nuclei or nukleoulus child. In the cytoplasm of animal cells, there are several organelles such as ribosomes, mitochondria, lysosomes, smooth and rough endoplasmic reticulum, Golgi apparatus / Golgi apparatus, centrioles, peroxisomes. Animal cells, such as animal cells described in the above sense, does not have a cell wall but has called the extracellular matrix sheath cells.
The structure of animal cells and organelles organelles animal cells
The structure of animal cells and organelles organelles animal cells
The function of the animal cell organelles mentioned above are described as follows:
Mitochondrial function
Mitochondria function as "energy factories" for animal cells. ATP as an energy animal cells and many other cells. With the mitochondria, a nutrient that has been processed or diglikolisis in the cytoplasm as anarobik process will be entered into the mitochondria as acetyl co-A and then with the help of oxygen will be perfected in the mitochondria. Read more details about this in the article organelles mitochondria and mitochondrial function
Endoplasmic reticulum function
The endoplasmic reticulum is a strong role in the synthesis of substances or molecules needed by animal cells, especially for cell regeneration and cell growth and development. Smooth endoplasmic reticulum plays an important role in the synthesis of a lipid, or fat, while the rough endoplasmic reticulum (with the help of ribosomes) play a role in protein synthesis. For more jelaskanya about animal cell organelles structure of this one and its functions, please read the article on the endoplasmic reticulum and functions
Function of the Golgi Apparatus / Golgi Apparatus
Cell organelles this one looks like a bearing with a grain of granular vesicles around. Golgi body itself has the functions related to protein synthesis and cooperate with the endoplasmic reticulum and ribosomes, besides, as organelles that function in the maintenance of animal cells and other cells (which have) to deliver the required substances using mikrovesikel. Moreover, the process of exocytosis in animal cells occurs in the presence of this Golgi body. For more details about the Golgi body and its functions, please read the article Golgi Agency
The function of lysosomes
Lysosomes are organelles animal cells which have a unique structure that has a single membrane organelles that function as a "cleaner" animal cell cytoplasm. Lysosomal size of about 0.25 um to 0.5 um hydrolytic enzymes in the enzyme-containing organelles her to be able to digest a variety of substances composed of proteins, fats and carbohydrates even phosphate. With the help of lysosomes, animal cells can destroy the substances are difficult to decompose, recycle damaged organelles. For more details, read the article structure and function of lysosomes.
peroxisome function
Peroxisomes are organelles found in all eukaryotic cells. Peroxisomes have a single membrane similar to lisosom.Organel cells containing approximately 50 of these enzymes help in the process of oxidative animal cells. In animal cells, peroxisomes play a role in the reform process fatty acids and cell toxins such as alcohol and H2O2 at the same time by using the enzyme catalase. One important function of peroxisomes for animals is as a catalyst in the first reaction plasmalogen formation needed in the formation of nerve cell myelin.
The function of centrioles
The centrioles are organelles animal cells that play an important role in the process of cell division animals. Through centrioles, yarn division will appear (spindle) that separates the homologous chromosomes so attracted towards the centrioles that has been split as well (at the poles, respectively). Read more clear about the role of centrioles in animal cells mitosis
In addition to the above cell organelles, cytoskeleton still having the structure of the skeleton that keeps the shape of animal cells. Although not as an animal cell organelles (because it is located outside the cytoplasm), animal cells also have flagella that animals play a role in cell motility (movement) of sperm cells in particular instances. In addition, there is also mikrovilis (mikropili).
Definition and structure of plant cells
Drafting a variety of plants that you saw yesterday and today, is composed of cells of plant cells which then form a network of plants. Understanding plant cells are cells that have protoplasm, cell membrane and cell wall composed of cellulose (or hemi-cellulose) and chloroplasts in the cytoplasm so that it can carry out photosynthesis. Understanding plant cells from the other side is a cell that is capable of forming his food and energy with the help of light energy or photoautotrophs with the help of chloroplasts and chlorophyll.
The cell structure of plants ranging from the outside, you can find their cell walls are thick but has little slits where the entry of substances needed, after all, it is their cell membranes. The cell membrane is the same with animal cell membranes, composed of a phospholipid bilayer. Then organelle organelles similar to animal cells will be found in plant cells (except centrioles which only exist in plant cells are motile). In addition, you can see the vacuoles in animal cells and many small but in plant cells generally only amounted to one and a large and located amid the so-called central vacuole. For more details about the structure and function of vacuoles please read the article on vacuoles.
The structure of plant cells and organelles of plant cells
The structure of plant cells and organelles of plant cells
To find out more about the structure of animal cells and plant cell structure, and the difference between these two cells, please read some of the articles below:
Animal cells: Structure and function of cell organelles animals
Plant cells: eukaryotic cells with cell walls
The cell membrane: structure, Authors and nature
Plant cell walls
Organelles and cell membranes
Differences in plant cells and animal cells in a table
Definition and Structure of a bacterial cell
Definition of simple bacterial cells are cells that are in a group prokaryotes. Understanding the bacterial cell is a cell that is walled cell but does not have a nuclear membrane and organelles except ribosomes and other organelles mesosom (organelles while).
Although the structure of the bacterial cell has a cell wall but different cell wall constituent of plant cell. The difference lies in the content of peptidoglycan in the cell wall of bacteria. Differences in the amount of peptidoglycan in the cell wall of bacteria makes it formed two kinds of bacteria are gram-negative and gram-positive bacteria.
Bacterial cells have a variety of forms ranging from cocci (round), rods (bacilli) and spiral. One thing you have to emphasize the bacterial cells are, bacterial cells do not have a nuclear membrane so that the cell nucleus dispersed in the cytoplasm. for more details, please read this article Bacteria.
The cell structure Protozoa: Amoeba Proteus
Amoeba Proteus is a unicellular animal cells. Why are categorized as animal cells? Because the amoeba has a characteristic feature of animal cells, namely motility, and cell structure it has. The difference with multicellular animal cell is an organ system that is owned by amoebas performed by organelles organelles that has, so there are differences in cell organelles organelles. For more details about the structure of this unicellular animal cells, please read this article Amoeba Proteus.
So explain the meaning of the cells, the structure and function of organelles as well as many kinds of cells in general. For further be explained about the understanding of the fungal cell and structure. That is all and thank you
- See more at: http://apapikir.com/2016/02/11/pengertian-struktur-dan-fungsi-dan-organel-sel/#sthash.72IUBTJj.dpuf
Function of the Golgi and Golgi bodies
TOPICS: Golgi Apparatus and Function of the Golgi Apparatus
Function of the Golgi and Golgi bodies
Function of the Golgi and Golgi bodies | This time belajarbiologi.com will explain about the Golgi apparatus and the function of the Golgi apparatus. Ade ade need to understand that the Golgi body or Golgi apparatus is one of the cell organelles.
Let us know the history of the discovery of the Golgi Apparatus or Golgi Apparatus prior to the Golgi body parts and functions.
History The discovery of the Golgi Apparatus
Golgi bodies officially discovered in 1898 by a nerve disease bernamaCamillio Golgi. Golgi find buildings in the form of dark woven on a sample of brain tissue cells which had previously been fixed into a solution of salt staining bikhromat with Ag.
Previously, the Agency Golgi or Golgi apparatus named by the inventor by the name of Apparatus reticularis then subsequently to honor Mr. Camillio Golgi, generation after replacing into the Golgi Apparatus. After the discovery of the electron microscope, the Golgi apparatus and then called the Golgi complex or Golgi region.
At first only attributed to nerve cells, but after a few and after a series of studies, it was found that the Golgi apparatus is an organelle that exist in eukaryotic cells.
Several studies carried out to determine the characteristics of the Golgi body, and one of them is color or use H.E. staining Golgi body, Golgi body turns tida own items to the ribosome so it does not show color or just show a clear area.
Structure and Form Golgi Apparatus
See the Golgi bodies and look at the picture below
Golgi apparatus: Structure and part of the Golgi body
Golgi apparatus: Structure and part of the Golgi body
Golgi body like you see above, it looks like bearings are plated and surrounding bubble Golgi. Bubbles in the Golgi body is divided into three, namely:
1 Sakula which is bubble shaped like flattened discs are arranged piled stacks each separated by a narrow gap. Being both surfaces of each Sakula not the same, namely one another convex and concave surfaces. In Sakula located near the top of the cell, the edge Sakula partially visible bubbles are released into items to sekreksi. Even bubble also released from the concave surface of the Sakula.
Secretory vesicles, secretory vesicles bubble is a bubble that is part of Sakula located on the edge
Mikrovesikel or transfer vesicles are micro bubbles with a diameter of approximately 40nm bergerak.Tolong note that mikrovesikel comes from the rough endoplasmic reticulum are released, but mikrovesikel has lost granules granules chromosomes. Mikrovesikel will unite with Sakula in the end.
How it Works Golgi Apparatus
Organelles Golgi apparatus or Golgi apparatus to work closely with the endoplasmic reticulum. RE will synthesize protein, then after protein synthesis occurs, will be formed vesicles subsequently transfer or mikrovesikel that dihantar to Golgi bodies organelles exactly on Sakula. So there will be a merger between protein dihantar with vesicle protein transport from the ER.
The first alleged may be enlargement of the Golgi body, but it does not happen because of the Golgi body or Golgi apparatus to form the secretory vesicles that functions in transporting the contents Sakula toward the top of the cell to outside the cell below.
In the Golgi apparatus or Sakula, compaction or condensation occurs, before dihantar into secretory vesicles. In the Golgi apparatus, there are 3 types galaktosil transferase enzyme that plays a role in repairing damage to the membrane protein due to differences in oligosaccharides.
Thereafter, the secretory vesicles will be out of the cell across the cell membrane via exocytosis. This process also improve the condition of existing cell membrane.
Function of the Golgi Apparatus
Based on the explanation above can be some function of the Golgi apparatus, namely:
1 .Berperan in improving the structure of the cell membrane or plasma membrane 2 2
2 .Berperan active in processing proteins or synthesized by the endoplasmic reticulum
3 .As organelles play a direct role in the transport of the material metabolism
4 .In the circumstances there was an error in the assembly of carbohydrates, may act as an assembler of carbohydrates, especially oligosaccharides on membrane proteins
5 .As organelles that play an important role in the synthesis of certain proteins protest
6 .Berperan important in protein sorting of certain proteins such as forming lysosomes
7 .Ikut in the formation of lipoproteins along with RE. smooth.
Thus the article about the Golgi apparatus and the function of the Golgi apparatus. Do you have learned only too well these days and understand biology. Thank you if they have to pay close attention of the articles of the Golgi body.
Metamorphosis Perfect and Imperfect
TOPICS: Metamorphosis Perfect and Imperfect
Metamorphosis Perfect and Imperfect
Metamorphosis is a biological process in animal development that involves changing the physical structure after birth or hatching (hatching). Metamorphosis is two complete metamorphosis and incomplete metamorphosis (Holometabola and Heterometabola). Here's a brief explanation of metamorphosis and incomplete, especially in insects.
Perfect Metamorphosis (Holometabola)
In complete metamorphosis, insects in the life cycle experiencing striking changes in the outside design and various organs of the stage. Insect larvae and adult forms of these groups no resemblance In complete metamorphosis. Changes occurring form are as follows:
Eggs -larva - pupa (chrysalis) - Imago (adult)
The eggs hatch into larvae. Larvae generally experience molting four times forming stage larvae stage larvae of one to four. In the larval stage. generally very active insect meal.
Just look at the larval stage of butterflies are caterpillars, which feed on the leaves very active. 3-stage larvae pupa stage four turns of the traffic system. In the pupa stage. inactive insect eating (fasting period). but the metabolic process remains ongoing. After experiencing growth and cell division, differentiation and organogenesis. then the pupa will turn into an adult insect (Imago). Besides metamorphosis, repetition of the process as well as on growth and embryonic development hlngga larvae eventually turn into the adult form.
So what caused the changes in the metamorphosis of insects? What happens to the body in the form of insect larvae that had been filled with food after metamorphosis? When an insect pupa form, occurs ProRes histolysis or destruction from within the network or the insect's body by oneself. Fluid that was previously used to digest the food used for the histolysis. In addition, cells called histoblast larvae will reorder the insect's body becomes an adult body use the nutrients in food and the larvae are ingested into the liquid in the pupa. The metamorphosis of insects in the pupa must not leak and a disruption means to interrupt the implementation of the histogenesis or the rearrangements that occur.
Examples of insects that undergo complete metamorphosis, among others butterflies, flies, mosquitoes. bees and beetles.
Metamorphosis Perfect and Imperfect
The process of metamorphosis, butterfly
Metamorphosis is not perfect (Heterometabola)
In incomplete metamorphosis, insects undergo metamorphosis from egg to adult is not prominent in the life cycle. Pradewasanya larval form or shape is called a nymph. Nimfa has similarities with the adult form (imago), except the reproductive organs and wings. Reproductive organs and wings on nymphs undeveloped. Only after the turn into an adult insect. Developing reproductive organs and insects can reproduce. In incomplete metamorphosis is not formed pupa stage. In sempurna.perubahan metamorphosis occurring form are as follows: Eggs - nymphs - imago (adult)
Examples of insect metamorphosis sempurnaa not include grasshoppers, cockroaches (cockroach), and the Crickets.
Thus a brief explanation about the metamorphosis and incomplete, I hope you learn also related posts below.
Metamorphosis Perfect and Imperfect 1
Incomplete metamorphosis grasshopper Melanoplus atlanus and stage of incomplete metamorphosis stage grasshopper
Biotic and abiotic
TOPICS: Biotic and abiotic
Biotic and abiotic
Biotic and abiotic
Symptom of biotic and abiotic
In a simple biotic is all part of the environment that consists of living things such as animals, plants and fungi. While abiotic is all part of the environment which shall be composed of inanimate objects such as rocks, soil, water, air and others.
ecosystem
In Biology discussion on biotic and abiotic usually given for the ecosystem. Why is that? Since the definition of the ecosystem is the venue for interksi between living beings (biotic) and liingkungannya (biotic). Well, so the condition of the formation of the ecosystem are twofold factor biotic or living creatures, and there is abiotic or environmental factors. Both these factors can not be separated because it is a unified whole. Living things need to survive abiotic factors, such as animals need water to drink, took land for tinggan and need a stable temperature to survive.
examples of Ecosystem
As an example of a very large ecosystems can be found in the environment around us, ranging from home ecosystem, paddy ecosystems, coastal ecosystems (mangrove) and others.
Try to observe the environment around the house, you will find some animals such as cats, caterpillars, ants, ornamental plants, and a variety of other living creatures. The living creatures including into biotic components. In addition to living beings, in around your house there are also objects or components that are not life called abiotic kompenen. Examples of such abiotic components of earth, stone, oxygen, carbon dioxide, air, temperature and sunlight. Within this ecosystem components of biotic and abiotic components perform reciprocal relationship (interaction) so as to create a natural balance.
When animals eat plants or animals eat other animals, then there was interaction between living beings and other living beings, this is not an example of the ecosystem. While the plant absorbs water to perform photosynthesis is an example of the interaction between the components of abiotic or biotic components can also be called an ecosystem. Another example of an ecosystem is an ecosystem. Eating fish in the pond microorganisms (very small animals) and require water for life.
Thus, each component of the ecosystem often interact to form a working unit to form the balance of nature.
Viewing Ecosystem
To understand more about the ecosystem materials, biotic and abiotic you can make observations. Choose one of the small neighborhood around your home, such as home parks, gardens, pools or other places that there are plants and animals. Observe and record what is contained in these environments, ranging from the visible to the eye until that is not visible to the eye, such as air. Kelompokanlah what you wrote it into two groups, namely the biotic and abiotic group.
So hopefully post about the ecosystem, biotic and abiotic can add to your understanding. Thanks.
Metabolic Acidosis: Symptoms, Causes and Treatment
Metabolic Acidosis: Symptoms, Causes and Treatment
TOPICS: Metabolic Acidosis: Causes Symptoms And Treatments
Metabolic Acidosis: Symptoms, Causes and Treatment
Metabolic acidosis refers to a condition in which the body's acid-base balance disrupted due to increased acid production or decreased excretion and decreased production of bicarbonate. This condition can eventually lead to acidemia or acidity of the blood, in which arterial pH drops below 7.35. If this condition is not treated, it can affect the central nervous system and lead to coma and even death.
Acid is usually produced as a byproduct in a number of activities including the breakdown of fat metabolism. Normal balance between acids and bases managed by bicarbonate, which neutralizes the acid, thereby preventing excessive accumulation of acid. Therefore, the factors that contribute to one of excess acid production or decreased production of normal bicarbonate may cause metabolic acidosis.
Cause
Metabolic acidosis is caused by increased levels of acid due to increased production of acid or acid digest or substances that can be converted into acid.
It can also be caused by:
The loss of bicarbonate such as in cases of diarrhea and renal tubular acidosis.
Ingesting large amounts of substances such as antifreeze, aspirin, etc. It can also increase the level of acid in our body.
The accumulation of lactic acid is another contributing factor. This usually occurs because of the availability of sufficient oxygen in the metabolism of carbohydrates, such as in the case of heart failure and shock.
Malaria can also be responsible for this condition by destroying red blood cells and thus reduce the level of oxygen in the body. This in turn results in the accumulation of lactic acid due to anaerobic breakdown of glucose or carbohydrates. Acidosis type known as lactic acidosis.
The metabolic abnormalities can cause acidosis. The use of fat rather than carbohydrates, to reduce energy, such as in the case of diabetes mellitus, may lead to excessive production of acid. The breakdown of fat produces ketones and increase the level of acid in the body. This condition is known as diabetic ketoacidosis.
Sometimes, such as pH imbalance can occur even without the excessive production of acid. For example, in the case of people suffering from kidney failure, the kidneys may fail to remove the acid in the urine.
Conditions such as alcoholism and malnutrition are also associated with this condition.
symptom
Although metabolic acidosis is usually characterized by rapid breathing, symptoms may be nonspecific, and may vary depending on the underlying cause. However, some common symptoms are:
Chest pain, headaches, heart palpitations, muscle and bone pain, muscle weakness, abdominal pain, etc.
Lactic acidosis is sometimes characterized by low blood pressure and anemia.
In diabetic ketoacidosis, patients may suffer from high blood sugar and dehydration. As this condition can affect the central nervous system, individuals may experience anxiety and progressive drowsiness. Nausea, vomiting, loss of appetite, and weight loss are some other symptoms.
In extreme conditions, can lead to severe complications such as fainting, coma, and seizures.
Treatment
Doctors generally perform blood tests such as blood gas analysis and a complete blood count to diagnose the condition. Treatment depends on the underlying cause. However, if the blood pH drops below 7.1, the conditions may require administration of intravenous bicarbonate to neutralize the acid.
In severe cases, dialysis may be necessary. Mechanical ventilation is also used to relieve breathing problems.
Monitor and control the factors responsible for causing this condition is the best way to prevent the condition from worsening. Like for example, control of causes such as diabetes can help control the disease in patients with diabetes. Metabolic acidosis is often a symptom of some serious diseases such as kidney failure and heart disease and diabetes. So the proper monitoring of the symptoms of this condition will be very helpful in preventing complications.
classification of Nematodes
TOPICS: Classification of Nematodes
classification of Nematodes
nematode worms examples
Nematodes consists of bebearpa order as follows:
Roundworm (Ascaris lumbrricoides)
Live in the human gut, is cosmopolitan (found in all places). The body wall is composed of a cuticle, epidermis and longitudinal muscle layers where there excretion channel lateral, dorsal and ventral nerve cord are connected by anterior nerve ring, rounded body shape long section tapered ends. body length of female worms ± 0.5 cm, while the male worms 1.5 to 2.4 cm, mouth terlatak tip of the anterior and is equipped with three lips, the body relatively more straight female worms, while the posterior end of the male worms had that roll, food in the form of food juices, the digestive tract is composed of the above: the mouth, pharynx, intestine and anus. Roundworm (Ascaris lumbrriciodes) causes the disease known as ascariasis. Example: Ascaris Prasit in the human intestine, the intestinal parasite Ascaris megalocaphala horse, suilae parasite Ascaris in the intestine of pigs.
The life cycle of roundworm, the fertilized egg free kealam out with feces. If the eggs ingested with food or drink, then in the intestines of these eggs will hatch into larvae, then grow into adult worms.
Hookworm (Anylostoma)
Live parasites in intestines of humans and many dijumapai in mining regions, especially in hot climates. These worms suck blood that can lead to death, the blood sucking, this worm mengeluakan anti-coagulant agents (hirudin) which prevents blood clots. Body length of approximately 1-1,5cm in humans. Examples hookworm Ancylostoma duodenale is that there are areas of tropical Africa and Asia and Necator americanus are found in America.
Life cycle of hookworms, the adult worm is ectoparasites in the human intestine. Worm eggs can come out with human feces, the fertilized egg will hatch and the larvae within a day aimlessly filariform. These larvae can penetrate the skin, with the flow of blood to the heart and lungs, after that go back to the intestines and grow into adults.
Pinworms (Enterobius vermicularis)
Living in the human colon, the body length between 9-15mm. at the time of laying eggs, worms toward the anus to obtain the oxygen needed for growth. Movement of these worms can cause itching in the anus. The body affected by this worm infection may contain worms 5000. Example: Oxyuris equi anal horse or donkey.
Filarial worms (Wuchereria bancrofti)
Filarial worms have arthropod intermediate host animals, for example mosquitoes, and the host remains the man in the lymph vessels. If the dead filarial worms clog the lymph vessels, it causes swelling or disease elephantiasis (elephantiasis). Microfilariae can enter the human body through the bite of the Culex mosquito.
Muscle worms (Trichinella spiralis)
These worms cause the disease trichinosis in humans, pigs or mice. The worms enter the human body through the pork undercooked. In the human intestine, the larvae multiply into young worms then become adult worms. To prevent infection by this worm, the meat should be cooked thoroughly to kill young worms.
Heterodera radicola
Live parasites on the roots of the plant, causing swelling in the plant.
Thus post today regarding the classification of biological nematode, may be useful for us all.
The parts of the Digestive System
TOPICS: Parts Of The Digestive System
The parts of the Digestive System
Source: http: //www.biologi-sel.com/
The digestive system includes the mouth, esophagus, stomach, liver, pancreas, small intestine and colon. Every part of the digestive system has a very important role. If one part of the digestive system is not functioning properly, the whole system can fail. This can lead to disease. Have you ever thought how every bite of food you digest? Or where exactly the food you eat? Well, many of you may not think about it.
Main part Digestive System
Mouth
This is the first part of the digestive system and also a very important organ in communication and respiration. Mouth called oral cavity. When the food into the mouth, it is broken into small pieces by chewing. Saliva helps the food softer and acts as a lubricant to help swallow. Saliva contains digestive enzymes that break down fat and some of the starch in the food we eat. The food was damaged, softened and swallowed is known as bolus. As the bolus enters the esophagus and then, stomach, this is where the digestive process begins.
Esophagus
A 25 to 30 cm long muscular tube that connects the mouth and stomach known as the esophagus or throat. The esophagus is one of the main parts of the digestive system, because everything we eat pass through this pipe muscles. This is commonly known as the food pipe. It is located in the throat between the stomach and the mouth. The food passes from the pharynx to the esophagus. The pharynx is located right after the mouth in the neck. The normal function of the esophagus is to bring bolus, other liquids and saliva from the mouth to the stomach where the digestive process actually begins. Transport processes that take solid food and liquids from the mouth to the hull automatically.
side
Stomach is 30.5 cm and 15.2 cm wide and hollow. Gastric location lies between the esophagus and the small intestine. The hull is the main part of the digestive system and is divided into four parts, namely the cardia, where the bolus enters first, fundus, which is the curve on the gastric corpus, which is the central part of the hull and the pylorus, the very bottom of the hull where food enters the small intestine. When food enters the stomach, the enzymes and acids start breaking the food and turn it into a semi-liquid form, which is then entered into the small intestine.
Heart
The liver weighs about 1.5 kg or more. It is the second largest organ in our body and is located on the right side of Gastric cavity behind the lower ribs. It is reddish brown in color and the largest gland in the human body. The liver plays an important role in detoxification. The bile produced by the liver aids in digestion and absorption of fats. It plays an important role in metabolic processes. It is very difficult to survive without a heart, because this is one important part of the body, without which the body can not function.
Pancreas
The pancreas is about 6 in length and is located in the rear of the hull It produces pancreatic juices and hormones, such as insulin, and enzymes, and help to deliver food to the small intestine. The pancreas helps break down fats, carbohydrates and proteins, so they can be used by the body. The pancreas also neutralize the acid in the stomach It produces insulin, a hormone, and absorb sugar from blood.pancreas help break down fats, carbohydrates and proteins. If not enough insulin produced by the pancreas, a person can suffer from diabetes.
Small intestine
The steps of the small intestine between 6 to 7 m in length and 2.5 to 3 cm, and is located between the stomach and large intestine. The small intestine is divided into three parts, namely the duodenum, the first section, jejunum or the mid-section, and the ileum, which is the bottom of it. In the small intestine, chemical digestion takes place. It protects the body against infection and secrete intestinal juice as well. The food is thawed in the small intestine into the large intestine, where absorption of water takes place.
Colon
The length of the large intestine extending up to 1.5 m in length. It absorbs water and salt, and helps in the excretion of solid waste materials. Waste or waste in solid form, such as the colon absorbs water from food. Colon maintain fluid balance in our bodies. Some disorders of the colon is constipation, diverticulitis, diarrhea etc.
Every part of the digestive system has a role to play in its own way. One should eat healthy to think better and work better. Proper nutrition is a must. The digestive system is also affected by the disease and to distance themselves from the disease, we must always take care of our bodies.





